Cancer Disease – what you absolutely need to know!
A cancer disease of solid tumors (90-95% of all cancer diagnoses) often follow the same basic principles and are therefore surprisingly similar but yet on a molecular level it is a very individual ailment. Therefore, one should know some biological basics for a fundamental understanding to discuss with your doctor about treatment options.
Cell death and renewal
Every second, about 50 million cells die in our body, but also as many are newly formed by cell division. This means that with 50 million new cells there are also some errors in copying the DNA. These cells are normally recognized and eliminated by the immune system. Most of these faulty cells cannot survive anyway, but a few can have such errors or mutations that switch off their “self-killing mechanism” (apoptosis) and do not let the cells die. This can lead to an uncontrolled growth of these cells and thus to a cancer disease.
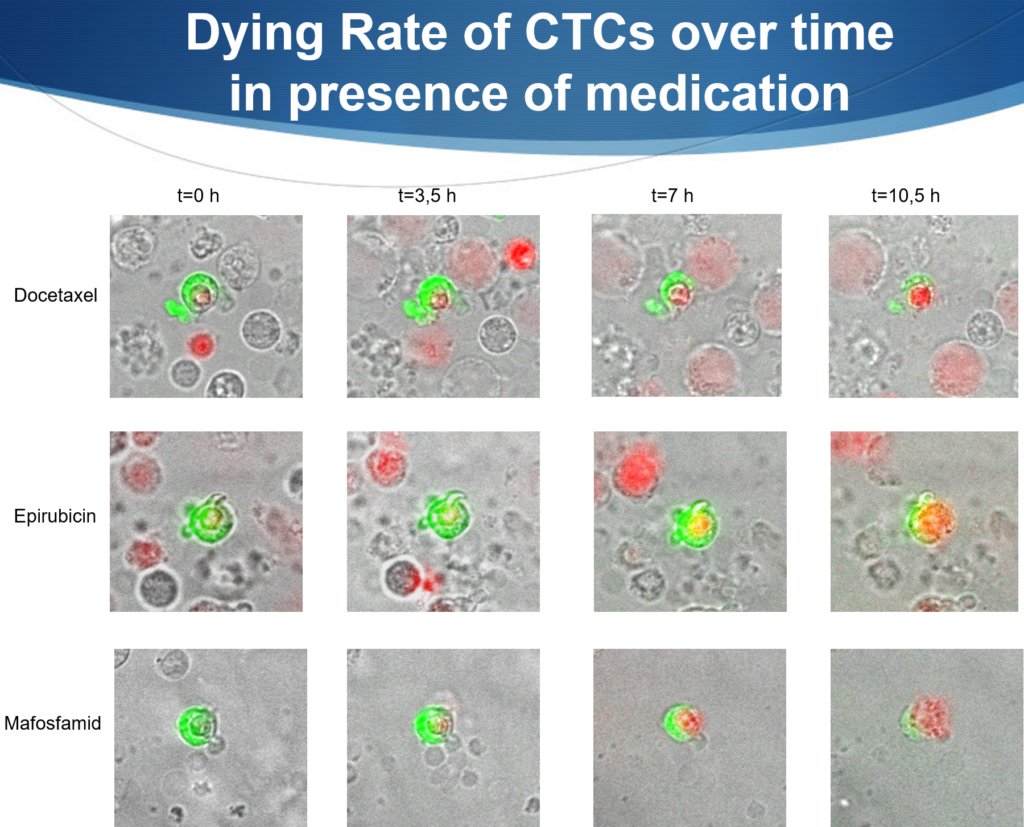
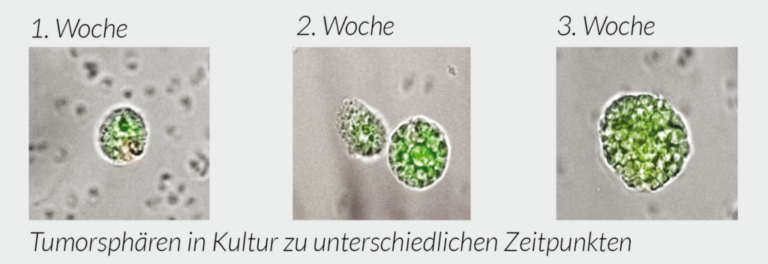
Drug testing on tumor cells (green). Here cancer cells are photographed over time with different drugs (Docetaxel, Epirubicin, Mafosfamid). The red color shows the staining of the cell nucleus (DNA) with propidium iodide. Red nucleus = dead cell, green staining = tumor cell (CTC) alive. In the background you can see normal cells that are also killed.
Tumor formation – Characteristics of a Cancer Disease
When a tumor forms, it starts from about 10,000 cells in size to release/seed 1 cell/day into the bloodstream. This does not sound like much, but if you consider that a tumor can only be seen by imaging methods from a size of about 0.5-1 cm in diameter, then this are already 3-6 billion cells. Theoretically, this means that at that time about 300,000-600,000 cells per day are scattered into the blood.
Significance of circulating tumor cells (CTCs) in the blood
These seeded cells are partly eliminated by the immune system but almost all of these CTCs are “sleeping” cells that can still be detected in the blood years later. They do not have the biochemical ability to settle in the body and form metastases.
However, there is a small percentage (about 0-12%) of these cells that have stem cell character, depending on the aggressiveness of the cancer. This means that they could settle in the tissue and form new tumors.
Structure of a Tumor
Tumors often have a very hard and dense tissue that can hardly be penetrated by arteria or veins. Therefore, the tumor often breaks down glucose without using oxygen to obtain energy (glycolysis). A tumor constantly “fights” for nutrients and energy to maintain its uncontrolled growth and harms therefore the rest of the body.
What are Solid Tumors?
Solid tumors are new formations of tissue that occur at a locally fixed site. They can grow anywhere in the body, such as in the breast, prostate, intestine, bones, lung etc.. Solid tumors can be benign or malignant. Benign tumors grow slowly and locally limited and do not form daughter tumors (metastases). Malignant tumors, on the other hand, destroy surrounding tissue and can seed cells into the blood. This can then lead to malignant new formations.
Treatment Principles for a Cancer Disease
Knowing the tumor characteristics like genetic mutations, receptors on the tumor cell surface, there are now different points of attack for a therapy of the cancer disease.
Chemotherapy in General
When a tumor is treated with chemotherapy, it is similar to peeling an onion. Layer by layer, cells are removed, killed or seeded dead or alive into the bloodstream. Chemotherapy is often quite unspecific and also targets other, healthy cells. It can have many different targets on and within the tumors.
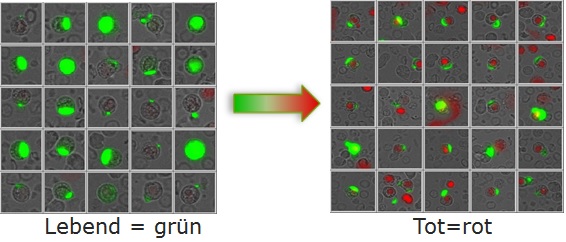
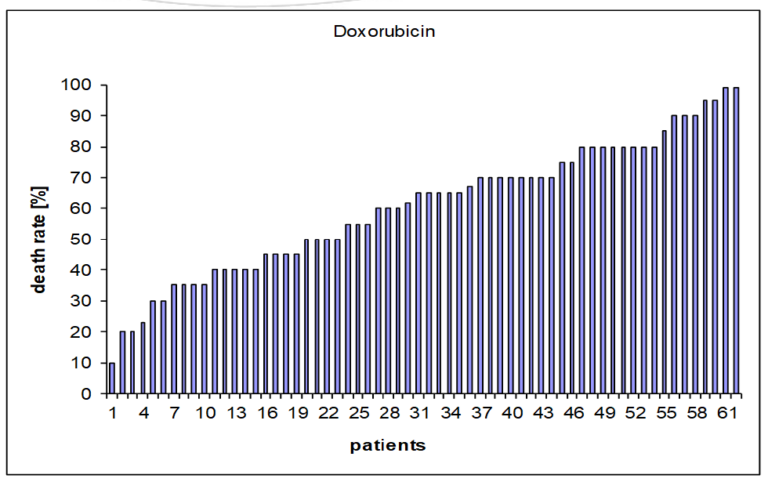
Checking the effectiveness of chemotherapy. How many percent of the green tumor cells will die.
Prevent Angiogenesis
Angiogenesis is the formation of new blood vessels. The tumor tries to promote the growth of vessels by releasing the VEGF signal molecule (vascular endothelial growth factor). With the drug Avastatin, one tries to bind the VEGF molecules to slow down the growth of vessels and therefore the tumor.
Blocking Receptors in a Cancer Disease
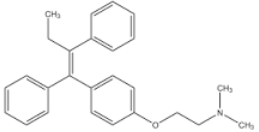
There are hormone-dependent tumors (breast cancer, prostate cancer, uterine cancer) that are stimulated to grow by hormones. This means that there are receptors on the tumor cell surface that promote growth of cells by binding a hormone (i.e. Estrogene). If you block these receptors (e.g. with tamoxifen or aromatase inhibitors) they no longer grow, or you prevent the formation of new hormones by drugs (aromatase inhibitors).
Antibody Therapy
Some types of cancer express certain proteins on the cell surfaces. Those can be bound quite specifically by antibodies. These marked cells can then be recognized and destroyed by the immune system.
DNA Damage
Other drugs (e.g. carboplatin) can embed themselves in the DNA within a cell and thus prevent the proliferation of tumor cells. Or the taxanes (e.g. paclitaxel) prevent the degradation of the “threads” (microtubuli) that pull apart the chromosomes during cell division.
Why do chemotherapies often not work?
Cancer is a very individual, unique disease. The diagnosed cancer has never existed on this planet and will never exist again, because too many genes (several hundred) are responsible for cell growth. And with each cell division, new mutations can occur.
If you look at e.g. a HER2-positive breast cancer, one patient has 10% of all cells with this receptor, but another patient has 90% of the cells positive. In the end, you treat 10% of the cells in one patient and 90% of the cells in the other patient. So it is always a kind of “fight” against statistics. And this is where the CTCs come into play.
What are circulating tumor cells (CTCs)?
Circulating tumor cells (CTCs) or circulating epithelial tumor cells (CETCs) are cells that have detached from a primary tumor and have entered the lymphatic vessels or circulate in the bloodstream. Since the primary tumors are epithelial tumors, they are called circulating epithelial tumor cells. Among the circulating tumor cells there is a subpopulation that is able to resettle in distant organs and grow new tumors, called metastases, which are responsible for the vast majority of cancer-related deaths.
The number of circulating tumor cells can vary greatly among patients. There is no threshold value that allows a specific prognosis or treatment recommendation. Studies have shown that even small tumors can have a high number of CETCs that are released into the bloodstream and vice versa.
What is the significance of circulating tumor cells (CTCs) for treatment?
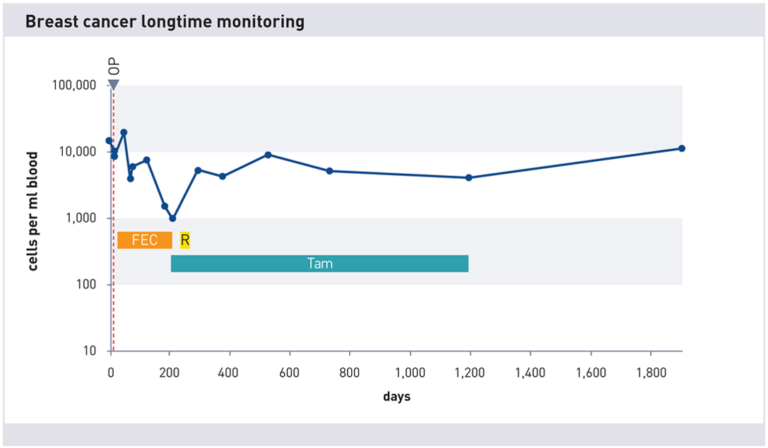
The detection and analysis of CTCs can help to assess the prognosis of patients at an early stage and find suitable, tailored treatments. CTCs can serve as markers for the response to a therapy by measuring their number before, during and after a treatment. A decrease in CTC number indicates a successful therapy, while an increase indicates resistance or progression of the disease. A change or adjustment of the therapy may then be necessary.
CTC Characterization for a Personalized Cancer Therapy
CTCs can also serve as a source of molecular information about the tumor by analyzing their genetic and epigenetic changes. These can change over time and under the influence of a therapy and lead to a heterogeneity of the tumor. The characterization of the CTCs can therefore provide clues to possible target structures for a personalized therapy or indicate acquired resistance to certain drugs.

These target molecules can be quantitatively determined in the laboratory and thus design an optimized therapy.
CTCs are the Better Alternative as Liquid Biopsy
CTCs are sometimes referred to as “liquid biopsy” because they can be an alternative or complement to a tissue biopsy. A tissue biopsy is often invasive, painful and not always possible or representative of the entire tumor. A blood sample, on the other hand, is easy and safe to perform and multiple samples can be taken over time to track the dynamics of the tumor.
What statistical aspects need to be considered when treating solid tumors?
The treatment of solid tumors is a challenge because it often does not reach or eliminate all tumor cells. This can have various reasons, such as:
- The tumor cells are spatially heterogeneous and have different accessibility for the therapy. For example, tumor cells in the center of a tumor may receive less oxygen and nutrients than those at the periphery and thus be more resistant to chemotherapy or radiation.
- The tumor cells are temporally heterogeneous and change over time. For example, tumor cells can acquire new properties through mutations or epigenetic modifications that give them a survival advantage or make them insensitive to a therapy.
- The tumor cells are functionally heterogeneous and have different roles and abilities. For example, some tumor cells can act as stem cells and renew themselves or differentiate into other cell types. These cells are called cancer stem cells and are considered particularly aggressive and quite therapy-resistant.
To cope with these challenges, we need statistical methods that take into account the heterogeneity and dynamics of tumor cells. For example, mathematical models can be used to simulate and optimize the growth, spread and response of tumors to a therapy. Or bioinformatic tools can be used to analyze and interpret the genetic and epigenetic data of tumor cells.
maintrac as Monitoring and Control Diagnostics
To know about the status of a cancer disease and whether adjustments of the therapy are advisable, a control of the cell number by means of the maintrac method is recommended. It can be started at any time with the tumor cell count measurement. If you decide to do so, there would be the possibility to order a blood collection set.
Conclusion
I hope I was able to give you an overview of the basic principles of cancer disease of solid tumors. Basically, it is always about a disturbed growth of the tumor cells. However, since there are hundreds of genes influencing this process, there are also accordingly many possibilities to attack the tumor more or less effectively. Therefore, it would always be advantageous to test before aggressive therapy measures whether the planned drug has any effect at all.
If you have any further questions, please feel free to contact me.
I remain with best wishes

Dr. Martin Burow, Biochemiker
info@DMB-Diagnostics.de